
Twelve years after my kidney transplant, it’s impossible to ignore both the freedom it’s given me but also one of its most serious challenges: skin cancer. With a family history of cancer and lifelong immunosuppression, I knew the risk was real.
After an initial diagnosis in 2021 and back-to-back diagnoses in late 2025/early 2026, I felt compelled to share my journey with biopsies, Mohs surgeries, and wound complications so others can benefit from my experiences.
Ultimately, here’s what I learned:
Stay vigilant about checking your body. Trust your gut. Don’t ever settle for appointments that seem eons away. While skin cancer is a common transplant experience, it’s valid to express displeasure if it happens to you. It may happen again. You can complain that time too. Your gratitude for a life-saving transplant is in no way diminished by being angry (or frustrated) you may have to endure skin surgeries, physical pain, stress, and medical bills. You’re not alone. The TransplantLyfe community is here for you.
This is my story…
——
Recently, when driving back home from an evening concert in New York City, I reflected on my health and all the gifts my kidney transplant has afforded me in the almost twelve years since my surgery.
The ability to enjoy a night out with friends, experiencing my favorite pastimes (live music) without having to worry about rushing home early to plug into a peritoneal dialysis cycler for 10 hours, is certainly at the top of that gratitude list.
However, the following morning, in stark contrast to the previous night’s fun, I woke up very early for a not-so-pleasant aspect of transplant life: addressing skin cancer (again).
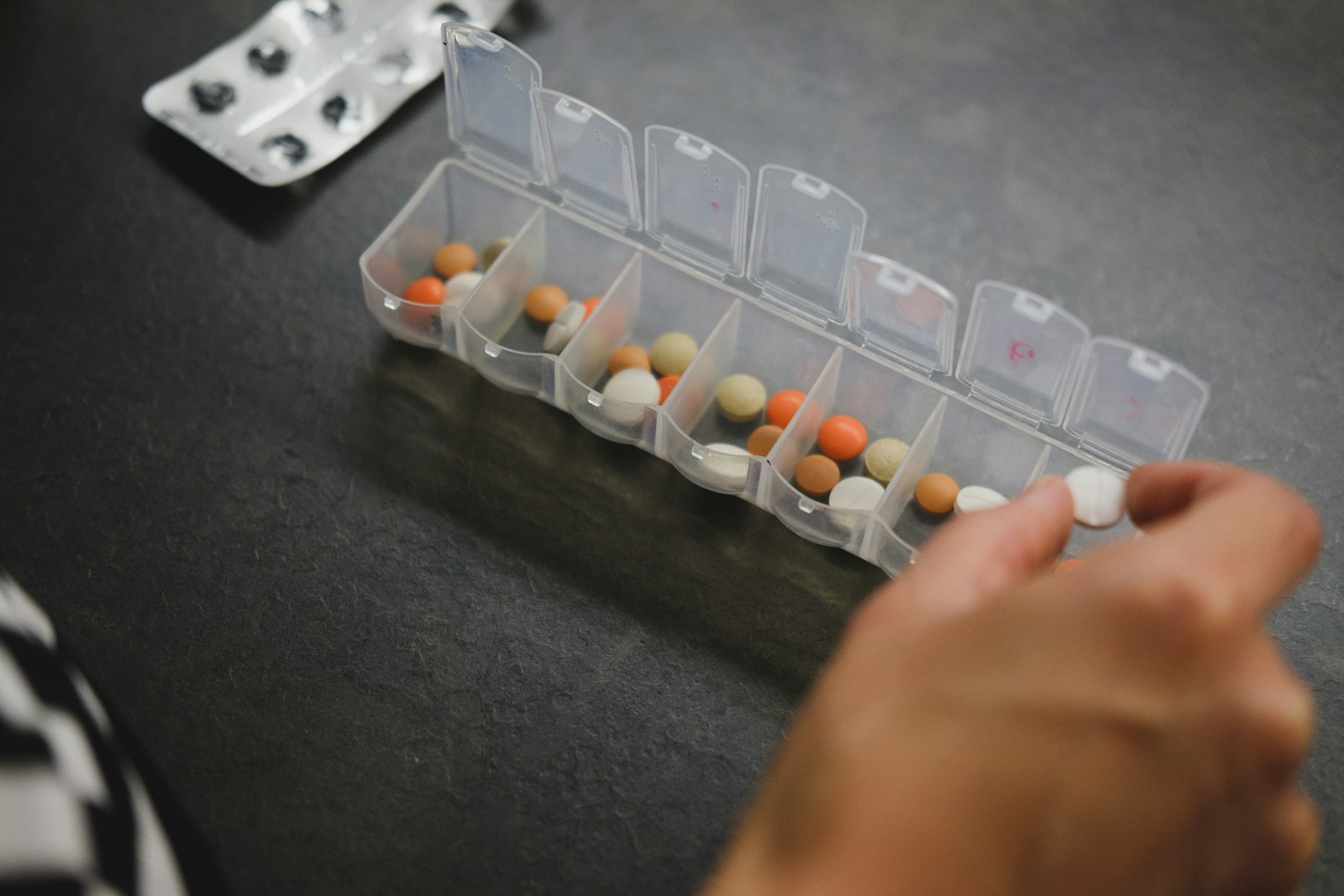
I’m currently 39 years old and my first experience with the dreaded “c”-word occurred in middle school when my father required invasive surgery to remove a melanoma on his arm. At the time, there was concern it had spread to his lymph nodes. Thankfully, the tumor was fully excised and Dad was OK. In the years since, he faced numerous instances of skin cancer, many of which involved Moh’s Surgery to resolve. (More on this highly effective removal technique later.)
When I received my living donor transplant from a close friend in 2014, I was told about the heightened risk of skin cancer due to the immunosuppressive drugs I would be taking for life. When factoring in family history, skin vigilance had to become an important part of my overall routine. Hats and high-SPF sunscreen became staples of my summer. I started seeing a dermatologist once a year for full-body screenings and tried my best to keep mental track of anything on my body that seemed off. Occasionally, some skin lesions such as actinic keratoses were frozen or burnt off just to be safe and to ensure these pre-cancerous conditions didn’t evolve.
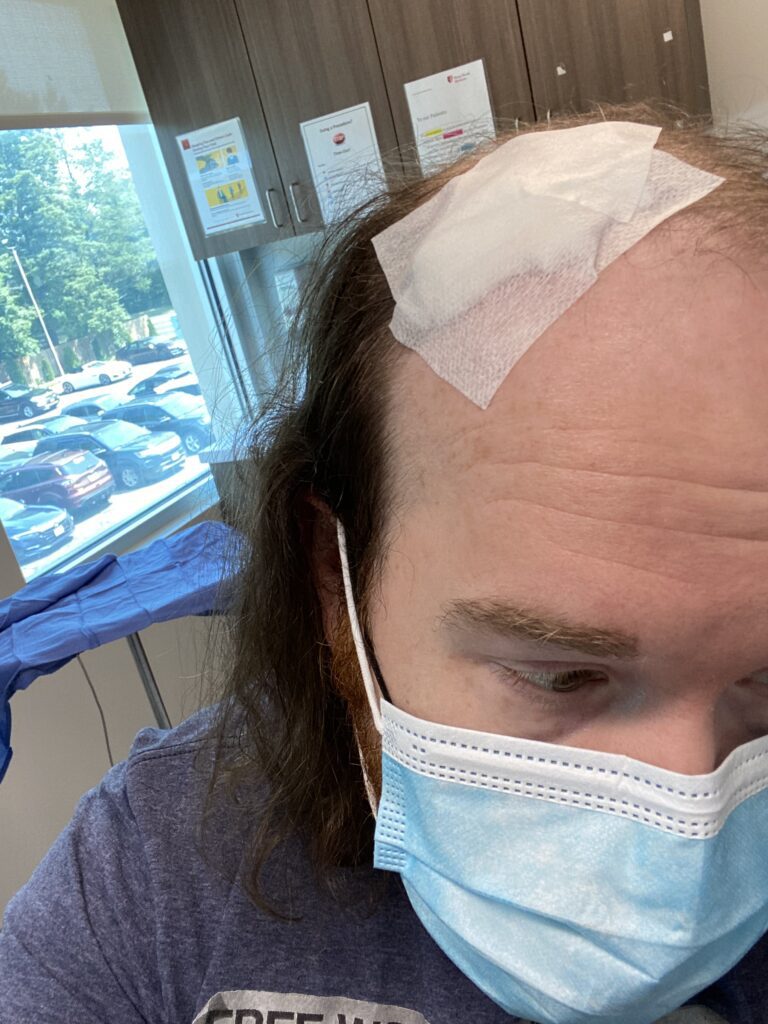
In May of 2021, 7 years post-transplant, I noticed a small, dime-sized mark on the front of my hairline. Initially thinking it was just dry skin, I applied cream and observed it over the course of a few days. Nothing changed and my gut felt that something was wrong. I called my dermatologist who unfortunately was booked out about 8 weeks. I took the next available appointment on June 28th, we biopsied the spot via a tiny surgical cut, and on July 4th weekend I received a call noting the sample was a basal cell carcinoma. My first-ever Moh’s Surgery was scheduled for the next morning, and I was told to bring a book or something to occupy my time as the procedure could take upwards of 3 hours.
Moh’s Surgery involves numbing the area with local anesthesia followed by extremely precise surgical removal of tissue, which is then viewed under a microscope to detect cancer cells along the edges. No cancer cells, no more surgery. If cells are present at the edges, it’s time for more cutting. The wound is commonly sutured closed or cosmetically restructured using skin grafts or synthetic skin substitutes, depending on the location and size of the gap. Moh’s is highly effective for skin cancer, with success rates as high as 99% according to Johns Hopkins Medicine.
While I was very grateful the cancer was gone, I was still annoyed it took me weeks to get a proper office visit. After doing some online research, I found another clinic, which was highly rated, took my insurance, and even offered night hours and same-day appointments. I never returned to my prior provider.
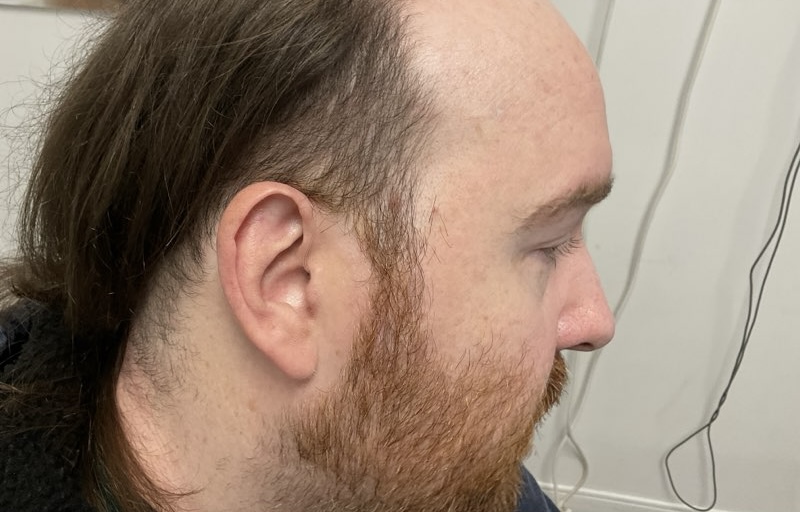
Fast forward to the Fall of 2025, when I noticed a small tan colored spot just above my sideburn. My current dermatologist had me come in right away; the spot was frozen, and a few weeks later, it was out of sight and mind. Until it wasn’t. By December, the spot began crusting again and a biopsy confirmed the presence of a basal cell. I opted for Moh’s right away, pre-holidays. I forgot how much the local anesthesia needles hurt when entering the skin. I was quickly reminded of how the fresh incision felt a few hours later once the meds wore off. The sutures on the side of my head were TIGHT and sleeping was very uncomfortable. On New Year’s Eve, we removed the sutures, closing the door on that situation, and randomly biopsied a bleeding spot on my leg, which I initially thought was a minor cut.
The first week in January 2026, I had a voicemail from the dermatologist about “the results” from my leg. Since they didn’t leave good news on my line, I assumed the obvious: more cancer. Sometimes I hate it when I’m right.
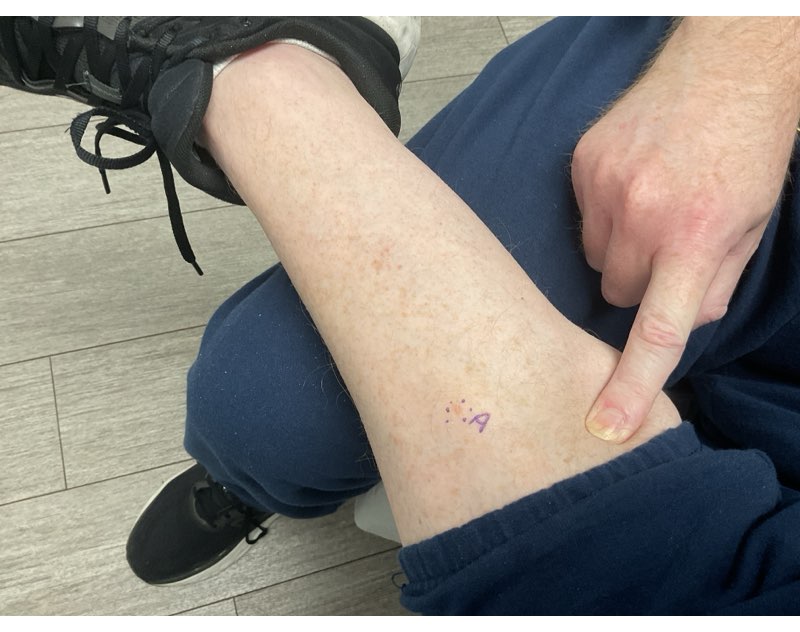
The leg biopsy site wasn’t healing well, likely due to me being immunocompromised, but after a few days of topical creams, on January 15th, I had Moh’s for the second time in under a month. This spot was on the shin of my left leg. Same painful needles, same rude awakening hours later when the numbing meds wore off. Sleeping was OK but standing up and feeling the blood flow to the wound was OUCH.

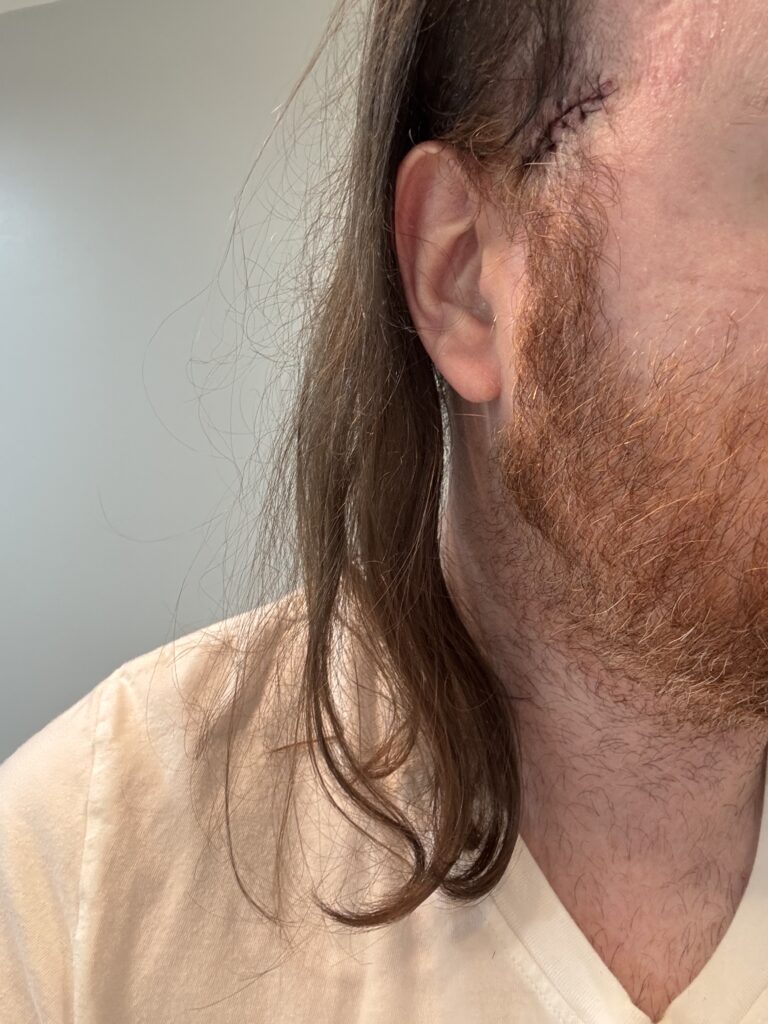
On January 18th, still reeling from the pain in my leg, I woke up to tons of blood on my pillow. Despite my head sutures having been removed weeks prior, the center of the wound reopened exposing a gaping hole. This is known as wound dehiscence. It can happen even with a perfect suture job due to a multitude of factors. Thankfully, no infection was present.
On January 19th, the wound was re-sutured, and I was back to bandages and the TIGHT pain. We agreed to leave these stitches in longer than usual and will do the same with my shin sutures.
While more cancer in my future is uncertain, all I know for sure is that I will be going to another concert next week.
“A Practical Guide to Achieving Your Health Goals” kicked off the TransplantLyfe University Webinar Series. Dr. Karin Hehenberger, Lyfebulb Founder and a kidney‑pancreas transplant recipient, was joined by registered dietitian, Maiya Hogan, MS, RDN, CD, CNSC. Together, they explored why setbacks are common, especially in the transplant community, and how reframing them can keep individuals motivated rather than discouraged. The session equipped participants with practical strategies to set achievable goals, stay realistic, and confidently adjust or level up their goals over time.
As I sit and write, it’s been five years in the chair—five years of waiting. Three times a week, on Mondays, Wednesdays, and Fridays, I wake up while it’s still dark outside to head to dialysis, my life-saving treatment. As my blood is cleaned and fluid is removed from my body, I sit and ponder: Will I ever get the chance to live a full whole life?
I’ve learned, with the help of my family and friends, that I must keep advocating for myself while also learning patience. Waiting comes with many emotions: hopelessness, depression, and frustration, and these are very real. I’ve given myself permission to feel them and own them.

I have worked tirelessly over the last two years, researching and studying different approaches to hopefully find myself a new kidney. I’ve learned that there are transplant centers that specialize in helping patients who struggle to get a kidney due to high antibodies. These antibodies can develop if you’ve had a previous organ transplant, a blood transfusion, or, for women, a past pregnancy.
Some centers try to lower antibodies using medicines or treatments such as plasmapheresis or IVIG. IVIG involves giving patients a concentrated dose of antibodies collected from donated blood from thousands of healthy people, and it remains the current standard of care. Other centers are exploring newer approaches, such as Belatacept and additional drugs. There is also discussion about xenotransplantation of organs or tissue from one species to another. Right now, NYU Langone and Massachusetts General Hospital are beginning early human trials.
In February of this year, I had the opportunity to speak with the team at NYU Langone. After some testing, I was able to get listed and participate in a clinical trial aimed at lowering my antibodies. Although it didn’t work for me, the experience was eye-opening and gave me valuable insight into how clinical trials operate. It also reinforced how important it is for patients’ voices to be heard in these studies.
For the trial, I worked with NYU for three months, traveling once a month from June 2024 through August 2024. Each time, I would fly to New York, receive the infusion, stay for 24 hours of observation, and then return home. After those three months, I followed up with local bloodwork until the study concluded in January 2025. In February, I had the opportunity to go back to NYU and meet with one of their teams to discuss the potential of a pig kidney. As of this writing, I am still waiting.
As I’ve learned from the great children’s book writer Mo Willems in his Elephant and Piggie book, Waiting Is Not Easy! I’ve had to remind myself to keep pressing on and look for new opportunities while waiting for a response from the NYU team. My fiancée, Tamiera, did some research online and discovered that the University of Chicago has a unique clinical trial using Belatacept and other drugs. I flew to Chicago, went through the process of getting listed, and met with a doctor who is willing to work with me. I am now wait-listed with them as well and continue to wait for insurance approval for the Belatacept treatment.
Sometimes waiting has meant I haven’t been the most available parent. I’ve missed my son Zander’s soccer games and practices, and my daughter Abby’s band and jazz concerts. That feels awful. At times, I’ve even felt like I’ve “wasted” my time waiting instead of being fully present in the here and now.
Recently, my family and I started a nightly meditation ritual using an app called The Way. It’s been teaching me how to be present in my body and physical space. It isn’t easy; anxiety still creeps in, and I worry. But I’ve learned that anxiety is normal. The key is not to stay stuck in it, but to notice it, then bring myself back to the present through something tangible around me.

As I continue to wait for a second kidney transplant, I want to remind others: you are not alone on this journey. While you wait for your call, look for ways to connect, share a meal with a friend, call someone, or send a kind message. Connection can make the waiting lighter.
Here at TransplantLyfe, we have a wonderful community of patients, caregivers, healthcare providers, and physicians, all here to support one another. I’m learning to wait not just to live my best life, but to live a life worth living—right here, right now.
Thanks for reading. Visit us at transplantlyfe.com to learn more from our community.
In Lyfe,
Alex

A chill is in the air and the pumpkins have taken over, it’s officially spooky season. As with many holidays that are centered on food, people can find Halloween hard to navigate especially if they follow special diets including post-organ transplant. If this is you, you might be asking yourself, or the internet, questions like, “What can someone with a transplant feel mostly good about eating during this time of year that won’t wreck my phosphorous and glucose levels?” or, “should I be eating dark chocolate, organic candy, or maybe lollipops to be healthier?”
You are not alone in your concern and confusion. Read on for tips to help get you through the maze of candy corn and chocolate bars to help you come out on the other side feeling good about your health and yourself.

Tip #1: Celebrate the Season
You’re not wrong if the first thing you think about during Halloween season is candy, and yet there are so many other ways to participate in the holiday beyond sweet treats. It may be helpful to write out a list of different activities to enjoy. For example:
- Visiting a pumpkin patch
- Pumpkin painting or carving
- Putting up decorations
- Dressing up in costumes
- Visiting a haunted house
- Organizing or participating in a Trunk or Treat for your community
- Walking through crunchy leaves
- Lighting a candle and cozying up with a good book or spooky movie
What would you add to the list?
Focusing on other aspects of the holiday may soften your concerns regarding food.
Tip #2: Don’t Get Tricked by Twisty Marketing
Food marketing is designed to make you feel like you must have a certain food, and you must have a lot of it, and you must have it right now. Otherwise, you’re going to miss out on a seasonal flavor, or you’re not enjoying the holiday the right way, and on and on.
The fact is, you need to choose what’s right for you. Asking yourself the following questions can help:
- Do you regularly experience GI issues?
- If so, you may want to avoid sweets marketed as “low in sugar” or “sugar-free” because that often means they are using sugar alcohols. Many people experience diarrhea when eating sugar alcohols. If you already have GI issues, these treats may make your issues worse.
- Are you trying to manage phosphate levels?
- It’s true that chocolate treats can be a source of dietary phosphate and hard candies like lollipops are not. If chocolate treats are your absolute favorite, that does not mean you need to say no, it means you need to think about portion control. Enjoying 1-2 fun-size candies a day will help minimize the impact on phosphate levels.

- Bonus tip – chocolate candy can be frozen to preserve the tastiness over time. Seal the candy in an airtight container and only defrost 1-2 pieces at a time.
- Are you trying to manage blood sugar levels?
- All candy – organic, milk and dark chocolate, etc. – will have an impact on blood sugar levels unless they are sugar-free. Just like with phosphate management, you can reduce the rise in blood sugar by portioning out your candy. It may be even easier during Halloween because most candies are already packaged into small servings.
- Regular physical activity, including walks, can also help your body use the sugar and other carbohydrates you eat throughout the day in a healthy way. This is a great time of year to be intentional about regular activity!
- Do you even like the sweets being offered?
- Sometimes we choose to eat foods we don’t even really like because of social pressure, seasonal scarcity, tradition, or for emotion management. When we take time out to ask ourselves if we’re even enjoying the experience, it can help us make healthier decisions.

Tip #3: Invite All Your Senses to the Party
Not to get too scientific, but research has shown that eating sweets in a mindful way, taking time to use all your sense to enjoy the treat, may increase your pleasure and reduce feelings of guilt.
The advice is to go all in when enjoying candy. That does not mean eating an entire bag of candy in one sitting. All in means really taking the time to enjoy and appreciate the experience of the treat using all five sense – hearing, seeing, touching, smelling, tasting.
If you want to practice, here’s a great resource on mindful eating for sweet treats: https://extension.purdue.edu/4-H/_docs/volunteer/resources-and-development/healthy-living/mental_health/mindful-eating-with-chocolate.pdf
Tip #4: Avoid Haunted Hauls
Long after the holiday is past, we continue to find little ghosts of Halloween haunting our house in the form of candy that hangs around too long. When we have candy lying around it might make us feel like we must eat the candy just to finish it off and not be wasteful. But there are alternatives to consider:
- Donate – local food banks, homeless centers, dentists’ office, military troops, and more may be willing to take unwanted candy off your hands
- Save it for later – storing candy in a cool, dry environment can extend it’s shelf life meaning the candy can be used for other holidays. Think stocking stuffers for Christmas, Valentine’s Day gifts, and Easter baskets.
- Share it – take it to work or other in-person event so others can share in the enjoyment
- Make arts and crafts – make a candy mosaic with your kids or grandkids. Or save it for gingerbread house decorations.
- Throw it away – if you can’t access an alternative option for any reason, it’s okay to just throw the candy away to remove it from your environment.
This Halloween, you’re invited to try out any of the tips above and experience the holiday in a new way, one that focuses on the joy instead of the guilt, stress, and shame. As you play around with different tips, share your experience with your TransplantLyfe community. Happy Halloween!

What is Xenotransplantation?
Xenotransplantation is the process of transplanting organs, tissues, or cells from one species to another. Scientists are exploring the use of pig organs for human transplantation because pigs have organs that are similar in size and function to human organs. This research is helping to address the shortage of human donor organs.
Why Pigs?
Pigs have been chosen for xenotransplantation for several reasons: Their organ size and function are close to that of humans; they reproduce quickly, allowing for a steady supply of organs; and scientists can genetically modify pigs to make their organs more compatible with the human immune system. Additionally, pig-derived medical products such as heart valves, insulin, and skin grafts have been used successfully in humans for decades.

What This Means for Patients
Patients who are not eligible for human transplants due to medical complexity may benefit from xenotransplants in the future. If xenotransplantation becomes widely available, it could mean:
- More available organs for patients on the transplant waitlist.
- Potentially faster access to life-saving transplants.

Where Are Pig Organs and Tissues Already Being Used?
While full organ transplants from pigs to humans are still in the early stage, some pig-derived tissues and cells are already being used in medical treatments, including:
- Heart Valves: Pig heart valves have been successfully implanted in humans for decades and are commonly used in valve replacement surgeries.
- Skin Grafts: Pig skin is used as a temporary covering for burn victims while waiting for human skin grafts.
- Corneal Transplants: Pig corneas are being explored as a treatment option for human eye diseases.
- Pancreatic Cells: Pig-derived pancreatic cells are being researched for their potential in treating type 1 diabetes by producing insulin in humans.
A Brief History of Xenotransplantation
Xenotransplantation is not a new concept. Scientists and doctors have been exploring the possibility of using animal organs in humans for over a century. Here are some key milestones:
- 1905: French surgeon Mathieu Jaboulay attempted the first xenotransplants by using pig and goat kidneys in human patients. However, these early attempts failed due to immediate rejection.
- 1963: Dr. Keith Reemtsma at Tulane University transplanted chimpanzee kidneys into humans. One patient survived for nine months before succumbing to organ rejection.
- 1984: Baby Fae, an infant born with a fatal heart defect, received a baboon heart transplant. Unfortunately, she only survived for 21 days due to immune rejection.
- 1990s: Advances in immunosuppressive drugs and genetic engineering improved the chances of xenotransplantation success.
- 2010s: Scientists began developing genetically modified pigs to remove the sugar molecule Gal-1,3-Gal, which triggers human immune rejection.
- 2022: A genetically modified pig heart was successfully transplanted into a human patient for the first time. The patient survived for two months, marking a significant milestone in xenotransplantation research.
- 2024:
- March: Richard Slayman became the first person to receive a genetically modified pig kidney transplant at Massachusetts General Hospital. The kidney functioned well, though he passed away in May from unrelated causes.
- April: Lisa Pisano underwent a pig kidney transplant combined with a heart pump at NYU Langone Health. The kidney was later removed after 47 days.
- November: Towana Looney, a dialysis patient for eight years, received a pig kidney transplant at NYU Langone Health. Her kidney was removed after 4 months.
- December: eGenesis received FDA approval to perform up to three pig‐kidney transplants in humans under a Compassionate Use (Expanded Access) protocol. This clearance allowed them to proceed with individual transplants before starting formal clinical trials.
- 2025:
- January: Tim Andrews received a pig kidney transplant at Massachusetts General Hospital. The kidney lasted a record 271 before being removed. Andrews has returned to dialysis.
- February: United Therapeutics received FDA clearance to begin clinical trials on gene-edited pig kidneys, with the first human transplant expected in mid-2025.
- June: Bill Stewart received a pig kidney transplant at Massachusetts General Hospital. As of October 2025, the kidney is functioning well.
- September: A new eGenesis trial is approved to provide gene-edited pig kidney transplants to 30 people age 50 or older who are on dialysis and the transplant list. Another developer of gene-edited pig organs, United Therapeutics, is about to start enrolling people in a similar FDA-approved study.
- 2026
- January: Tim Andrews a received a human kidney transplant becoming the first person to receive one after a pig kidney.
Key Players in Xenotransplantation Today
Several institutions are leading the way in xenotransplantation research:
- Massachusetts General Hospital (MGH): Conducted the first pig kidney transplant in a living human (2024) and continues to perform pig transplant and clinical research.
- NYU Langone Health: Has performed multiple pig kidney transplants and continues clinical research.
- University of Maryland Medical Center: Conducted the first pig heart transplant in a human in 2022 and continues xenotransplantation research.
- Columbia University: As part of its Kidney and Pancreas Transplant Program, scientists are conducting research across immunology and xenotransplantation to achieve induction of xenograft tolerance.

The Biggest Challenge: Organ Rejection
One of the main obstacles in xenotransplantation is that the human immune system sees pig organs as foreign and attacks them immediately, a reaction called hyperacute rejection. This is mainly because pig cells have a sugar molecule called Gal-1,3-Gal, which human bodies recognize as a threat. Rejection isn’t just immune-based — coagulation (blood clotting) is also a key hurdle in pig-to-human transplants.
How Scientists Are Solving This Problem
Researchers have developed several strategies to overcome rejection:
- Genetic Modification of Pigs – Scientists have removed the Gal-1,3-Gal sugar from pig cells using gene-editing technology. This makes pig organs less likely to trigger an immune attack.
- Improving Immune Compatibility – By selectively breeding pigs with certain immune system traits, scientists have increased the success rate of organ acceptance in animal studies.
- Short-Term Immunosuppression – Just like with human organ transplants, medications can be used to temporarily suppress the immune system and help the body accept the new organ.

Where Are We Now?
As of 2025, several patients have received genetically modified pig kidneys that functioned without hyperacute rejection, marking significant progress. Xenotransplantation is still in the early stage, but progress is being made. Recent studies have shown that genetically modified pig kidneys and hearts can function in human recipients for extended periods. Researchers continue to refine the process to make xenotransplantation safer and more effective.

What’s Next?
Ongoing research is focusing on:
- Further improving genetic modifications to prevent rejection.
- Ensuring long-term survival of transplanted pig organs in human patients.
- Understanding how xenotransplantation might impact overall health and longevity.
- Immunology and tolerance to reduce (or eliminate) the need for immunosuppressive medication (moonshot goal!)

Final Thoughts
Xenotransplantation has the potential to revolutionize organ transplantation, offering hope to patients in need of life-saving organs. The goal is not to replace human donors, but to expand access and save lives where human options fall short. While it is not yet widely available, rapid advancements in research are bringing us closer to making it a reality.

“My name is Caroline and I have a heart transplant.” It was my answer to the question my professor had asked- “State your name and one cool fact about yourself for the class”. It was the first time I sat in a college classroom in over three years. I could feel my cheeks flushing pink when it came time for me to answer.

I am used to making speeches for crowds of adults, for other disabled people, and regularly speaking to at least one healthcare professional a week. But when I went back to college as someone who had beaten cancer with a port still in my chest, as a heart transplant recipient, and as a disabled person in a colorful wheelchair, I had no idea how new people my own age would receive me. I remember telling myself before the first day of school that I would not bring up all of the “complicated” stuff until people got to know me. I wanted to have an opportunity for people to see me, and not just see what I’ve been through.
There are so many other interesting things about myself, but I found myself saying “I have a heart transplant” as my “cool fact”. It was a split-second decision, I’ll admit that. I didn’t know why I chose that until later when it hit me- if I was going to truly start fresh, I didn’t want to hide a single part of me. No feeding into that ‘be the cool mysterious girl movie trope’ type (because what does that even mean). I am a very out there person; I find it near impossible to shrink to hide who I truly am. Though when I looked around the room after my statement, I saw the pairs of students sitting together, and in realizing that I sat alone, I worried that I had made a mistake.
At the college I attend, we have an accessibility counselor, who told me a few weeks into school about a group of students who were trying to start a Disabled Students
Union on campus. I went to the first meeting as Secretary of the union, again not knowing how I would be received. I was greeted by the most welcoming group of people who were so unashamed of being themselves. I immediately felt safe.
We all had such different disabilities, but we all bonded over commonalities like medications we had been on and our experiences of our lives being so intertwined with hospital stays and doctors. We ranted over viewpoints and labels we hated being imposed on us. They became the first college friends I made in nearly four years. I left each meeting feeling closer and closer to the person I always wanted to be after my transplant- shameless and loudly proud.
Over time, I became more involved with things like the Student Government and the Student Diversity Council. Each time I spoke in front of my peers, my cheeks got less pink and my voice shook less. My outfits got more and more colorful, I wore my blue heart-shaped glasses to class often, and my confidence in not only my transplant life soared, but also within my own voice. I wasn’t so afraid anymore. I noticed more of my peers would complement my wheelchair wheel colors that I would change out weekly, and it became easier to connect with strangers. By the time April rolled around, I decided I wanted to do something kind of bold.
April is National Donate Life Month, and I wanted to bring it to my campus. I organized a card drive, where cards would be decorated by students and sent to a hospital for transplant patients and donor families. I also decided it was time to put my voice to use. I enlisted the help of a close friend who has a heart transplant and a faculty member (who later also became a friend) that had donated a kidney to a stranger, to speak in front of students with me. To my surprise, students showed up with genuine curiosity; the event was a success. I felt so incredibly proud.
When I went back to school, I feared I would not make a single friend. That no one would care about transplant life. I was so thankful to find that people did care. I was not only accepted by my peers with open arms, but I made some of the best friends I
could ever ask for. My new friends make me transplant-friendly ice cubes in cute little shapes and always check my diet requirements before giving me food they made. It’s so simple, but it means more to me than they’ll ever know. My lifelong protector and best friend, Cassidy, would tell you that she was right- she knew the right people would find me. I even found some self-acceptance along the way. Next year, I will serve as the President of the Disabled Students Union, being voted in by my peers. I have so many plans on how to bring organ donation and disability awareness onto campus, knowing there are wonderful people who will care about it.
The entire experience of going back to college as a transplant recipient has taught me a few things. What I’ve been through is such a big part of me and there’s no shame in that. Doing it scared, even if your cheeks flush and your voice shakes, is something to be proud of. That self-acceptance is right around the corner if you stop shrinking yourself to fit a self-imposed limitation. But the biggest lesson of all? If you live in truth, if you live as who you truly are, the right people and the right path will always find you.


My name is Emmitt Henderson III, and my journey with Lupus has been anything but ordinary. Diagnosed over three decades ago, I’ve faced countless battles, but none more life-altering than the fight against kidney failure, caused by Lupus Nephritis, that has changed the course of my life. After enduring multiple hospitalizations, organ failures, dialysis treatments, and near-death experiences, I found myself on a mission, not just for healing, but for hope. That hope came in the form of searching for a living kidney donor.

Out of this complicated journey, I created Male Lupus Warriors Corp, an organization dedicated to empowering men living with lupus and chronic illness. I wanted to create a space that wasn’t available for me during my struggles. Through my platform, I’ve had the honor of advocating nationwide speaking at conferences, attending policy events like Rare Kidney on the Hill Day, and educating communities about organ donation and health equity. My advocacy has earned me awards and recognition, but the real reward is knowing that my story is helping others rise through their health struggles.

I’ve traveled across and outside the country to share my truth, connect with fellow warriors, and amplify the voices of those often overlooked. And now, I’m proud to say I’m also an author, using storytelling to leave a legacy of strength and transformation.
I didn’t know I had a story till I told my story and this is my space to reflect, educate, and inspire. This is how I turn my pain into power.
End-Stage Renal Disease, Medicare Coverage & Long-Term Financial Planning
In Part 2 of our TransplantLyfe webinar series, we take a deep dive into the financial realities facing people with kidney disease—particularly around Medicare eligibility, coverage decisions, and long-term planning after transplant. This 60-minute session features real patient case studies and expert insights to help patients, care partners, and healthcare professionals better understand how to navigate one of the most complex—but critical—aspects of transplant care.
The webinar includes expert commentary from Craig Pressley, LICSW, Social Work Program Leader at Beth Israel Deaconess Medical Center, and is moderated by Kevin Schnurr, a kidney transplant recipient and TransplantLyfe Community Manager, and Dr. Karin Hehenberger, a kidney-pancreas transplant recipient and President of Lyfebulb.
The session explores two distinct case journeys: Maria, a preemptive transplant recipient with commercial insurance, and James, a dialysis patient on the transplant waitlist. Their stories illuminate how Medicare enrollment decisions—such as when to initiate Part A and/or B coverage—can dramatically affect financial obligations, insurance coordination, and access to essential medications like lifelong immunosuppressants. The panel explains the 30-month coordination of benefits period, Medicare Part B-ID protections, and how to avoid common pitfalls that can lead to unexpected costs.
In addition, the webinar highlights multiple financial assistance options, including:
- American Kidney Fund’s Health Insurance Premium Program (HIPP)
- Medicare Savings Programs (QMB, SLMB, QI)
- The Extra Help Program for Medicare Part D
- Pharmaceutical assistance tools like NeedyMeds, RxOutreach, and GoodRx
Whether you are preparing for transplant, recovering from surgery, or supporting a loved one, this webinar provides essential education to inform decision-making, reduce financial risk, and improve long-term outcomes.
Learn more at TransplantLyfe.com and join our discussion forum to continue the conversation.
The iBox Scoring System is a novel composite biomarker endpoint predictive of long-term graft survival after kidney transplantation
On track to be the first qualified reasonably likely surrogate endpoint in transplant for use in the FDA accelerated approval pathway
FDA aims for a 10 month review of the Full Qualification Package
MOUNT LAUREL, N.J., July 30, 2025 /PRNewswire/ — The American Society of Transplant Surgeons (ASTS) and American Society of Transplantation (AST) announced today that the Transplant Therapeutics Consortium (TTC) received notification from FDA of the Reviewability of the Full Qualification Package (FQP) for the iBox Scoring System, a novel reasonably likely surrogate efficacy endpoint for kidney transplant clinical trials. The FDA Drug Development Tool qualification program aims to communicate the final decision within 10 months targeting April 2026. The Full Qualification Package is the final step in the FDA qualification process and iBox scoring system is the first transplant endpoint to reach this stage of qualification.
Improving the long-term survival of transplanted kidneys is an important area of unmet need for transplantation recipients. The current efficacy failure endpoint has typically shown non-inferiority of immunosuppressive regimens but the iBox Scoring System is proposed to be used to demonstrate the superiority of a new agents compared to the standard of care as a co-primary endpoint in pivotal drug therapeutic studies. As a reasonably likely surrogate endpoint, the iBox scoring system would be used in the Accelerated Approval Pathway at the FDA designed to allow for earlier approval of drugs that treat serious conditions and fill an unmet medical need.
“It’s not surprising that using a clinical trials endpoint that is nearly four decades old has been a significant impediment to the development of new therapeutics for maintenance immunosuppression and has contributed to the relatively stagnate long-term outcomes of kidney transplants. I applaud the FDA for working with the TTC to develop the iBox as a coprimary endpoint for clinical trials in kidney transplantation. We have recognized the strong push from the patient community for new agents that promote both longer survival of transplanted organs and improved safety and tolerability. I hope that this is the first of many collaborative efforts to validate and implement new drug development tools to achieve that end,” said Kenneth Newell, MD, PhD, Professor of Surgery, Emory University and Executive Committee Member of the TTC.
Paul T. Conway, Vice President and Chair of Policy and Global Affairs, American Association of Kidney Patients (AAKP) and a 28-year kidney transplant recipient stated, “For decades, bipartisan national policy priorities, across multiple presidential administrations and Congresses, have included transplantation and sustainable long-term transplant outcomes as a means for saving more lives, protecting livelihoods, and reducing patient and taxpayer burdens. Yet, due to policy barriers, the transplant drugs patients currently depend upon to avoid organ rejection and a return to the transplant waiting list have failed to evolve and pose serious risks to both organ recipients and grafts. AAKP is optimistic that recent FDA action on the iBox is a signal the agency is putting transplant patients first, accelerating transplant drug development and re-aligning internal FDA decision-making to better advance long-standing American policy priorities.” Conway previously served as the inaugural Chair of the FDA’s Patient Engagement Advisory Committee for medical devices.
“We celebrate this step toward a regulatory process that will allow for many more drugs to enter into the transplant drug development path. The patient community has been starved of new approaches to keep their precious grafts alive and thriving for a longer time, with fewer side effects and complications. As a person living with transplants derived from selfless individuals, I can speak for the transplant patient community in applauding this progress toward our goal of leveraging modern day science to achieve one transplant for life,” said Dr. Karin Hehenberger, President Lyfebulb, and a kidney and pancreas transplant recipient.
About iBox
The iBox scoring system is a composite endpoint that utilizes multiple clinically relevant features (estimated glomerular filtration rate (eGFR), proteinuria, anti-human leukocyte antigen donor-specific antibody, and kidney graft biopsy histopathology) at one-year post-transplant to predict 5-year graft survival. The iBox scoring system (NCT03474003), has been developed and validated in a landmark publication released in the BMJ by a team of researchers led by Professor Alexandre Loupy in Paris French NIH (Inserm), APHP and Université Paris Cité, comprising 7557 patients from 10 academic centers from Europe and the US and 3 Randomized controlled trials.
About FDA Accelerated Approval
The FDA Accelerated Approval Program is designed to facilitate and expedite development of drugs that treat serious conditions, and fill an unmet medical need based on a reasonable likely surrogate endpoint (RLSE) that is supported by strong mechanistic and/or epidemiologic rationale. The Qualification of the iBox would allow it to be used as a RLSE for accelerated approval of novel new immunosuppressants for kidney transplantation. The Accelerated Approval program has historically been successful advancing new innovative therapies for HIV and cancer and most recently promoted the introduction of 4 new drugs for IgA nephropathy, a chronic kidney disease with no approved therapies 5 years ago before the use of proteinuria as a surrogate endpoint.
About TTC
The Transplant Therapeutics Consortium (TTC) is a public-private partnership with the FDA founded in 2017 by the American Society of Transplantation (AST) and the American Society of Transplant Surgeons (ASTS). This collaboration unites the transplant community, industry leaders, and regulatory agencies dedicated to the development of innovative tools, methods and processes for facilitating advancement of new innovative therapies to improve the lives of solid organ transplant recipients.
This submission of the FQP was accomplished by employing the resources of the TTC members and engaging with FDA throughout the biomarker qualification process. TTC is supported through funding and input from the AST*, ASTS*, argenx*, Arkana Laboratories*, Bristol Myers Squibb*, CareDx*, Critical Path Institute, CSL, CTI Clinical Trial Services, Inc.*, Eledon*, European Society of Organ Transplantation, the U.S. Food and Drug Administration (FDA), Hansa Biopharma*, HUS Helsinki University Hospital, Immucor, KU Leunven, Memo Therapeutics AG*, Natera, National Institutes of Health, Novartis, Paris Transplant Group, Pirche, Sanofi*, Takeda*, Talaris Therapeutics, Thermo Fisher Scientific, The Transplantation Society, Transplant Genomics*, University of Manitoba, and Veloxis Pharmaceuticals*. Any groups that would like to join in this effort or have information or data that may contribute to further advances, can contact JoAnn Gwynn at jgwynn@myast.org.
*Denotes Current TTC Members
The Transplant Therapeutics Consortium (TTC) was launched in April 2017 and co-founded by the American Society of Transplantation (AST) and the American Society of Transplant Surgeons (ASTS). TTC brings together pharmaceutical companies, diagnostic companies, academic and nonprofit partners working toward a common goal of moving the field forward toward drug development solutions in transplantation.
American Society of Transplantation
Founded in 1982, the American Society of Transplantation (AST) is a non-profit, 501(c)3 organization dedicated to advancing the field of transplantation and improving patient care by promoting research, education, advocacy, organ donation, and service to the community through a lens of equity and inclusion. The society is the largest transplant organization in North America (consisting of nearly 5,000 professional members), representing a majority of the nation’s medical professionals engaged in the field of transplantation.
Learn more at myAST.org.
American Society of Transplant Surgeons
The American Society of Transplant Surgeons (ASTS) was founded in 1974 by 127 surgeons to unite the professionals who were advancing the fledgling field of organ transplantation.
From this small group of dedicated individuals, we have grown to represent over 2,000 transplant professionals who are dedicated to saving and improving lives through the transformative power of organ donation and transplant surgery. Learn more about ASTS by visiting our website at ASTS.org.
MEDIA CONTACTS:
Shauna O’Brien – AST: sobrien@myAST.org
Anna Shults – ASTS: anna.shults@asts.org
SOURCE American Society of Transplantation

When I joined TransplantLyfe, I expected to find information and maybe a few people who understood what I’d been through. What I didn’t expect to find was Jeanmarie, a best friend, a soul sister, and living proof that hope and strength don’t just survive – they thrive when shared.

We connected through our transplant journeys, each of us carrying a story marked by loss, healing, and the fight to keep going. But what truly bonded us was something deeper: a shared passion for advocacy, a fierce desire to turn pain into purpose, and the comfort of knowing that someone else just gets it.
From late-night check-ins to spontaneous New York City adventures, our friendship became its own kind of medicine. One of my favorite memories is the day we took a bike tour through Central Park. With the wind in our faces and laughter echoing between the trees, it felt like reclaiming life on our own terms. It was a celebration of every moment we’ve fought to live.

Whether we were shopping at Tiffany’s or simply breathing in the magic of the city, we weren’t just passing time; we were honoring it. We were honoring every heartbeat, every second chance, and every moment our transplants gave back to us.

Jeanmarie’s strength inspires me daily. Even on her hardest days, she lifts others up. Her voice fights for the unheard, and her joy reminds me that light still lives in the hard places. She’s the friend who remembers the dates that matter, who checks in without being asked, and who makes you feel seen in a world that sometimes overlooks transplant warriors.

Our friendship is a reminder that healing doesn’t always come through medicine or milestones. Sometimes, it shows up in a person who walks beside you, believes in your tomorrow, and never lets you forget that your story still matters.
So if you’ve ever wondered what hope and strength look like… It’s not just from survival, but from a connection.
By: LaVise McCray
